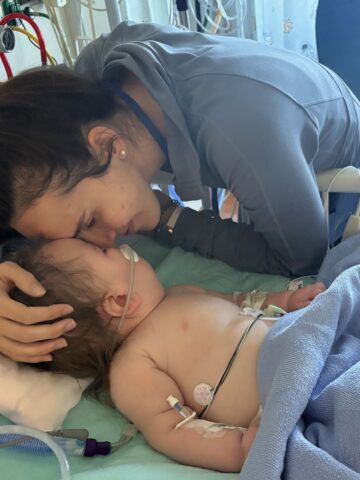
With time not on their side, CHOC care team pulls out all stops to save life of Emery Kline.
CureARS and CHOC symposium unites leading rare disease researchers
The event drew participants from representatives of eight children’s hospitals, 15 organizations, and a total of 200 attendees representing 20 states and four countries.
CHOC earns national distinction as Batten Disease Center of Excellence
CHOC is one of five Batten Disease Centers of Excellence across the country and the only one on the West Coast.
Improving outcomes and treatments for lysosomal storage disorders
Learn how the CHOC Foundation of Caring Lysosomal Storage Disorder Program and Metabolic Lab are advancing lysosomal storage disorder treatment.
CHOC receives prestigious NIH grant to study treatment for rare lysosomal storage disease
Grant’s support will help advance gene-therapy treatment for the rare lysosomal storage disease mucopolysaccharidosis type I (MPS I).
Rare mitochondrial diseases are the focus of Feb. 10 virtual CureARS event
The upcoming webinar is open to the public, and will share the latest developments in the battle against rare mtARS mitochondrial diseases.